I need help with homework
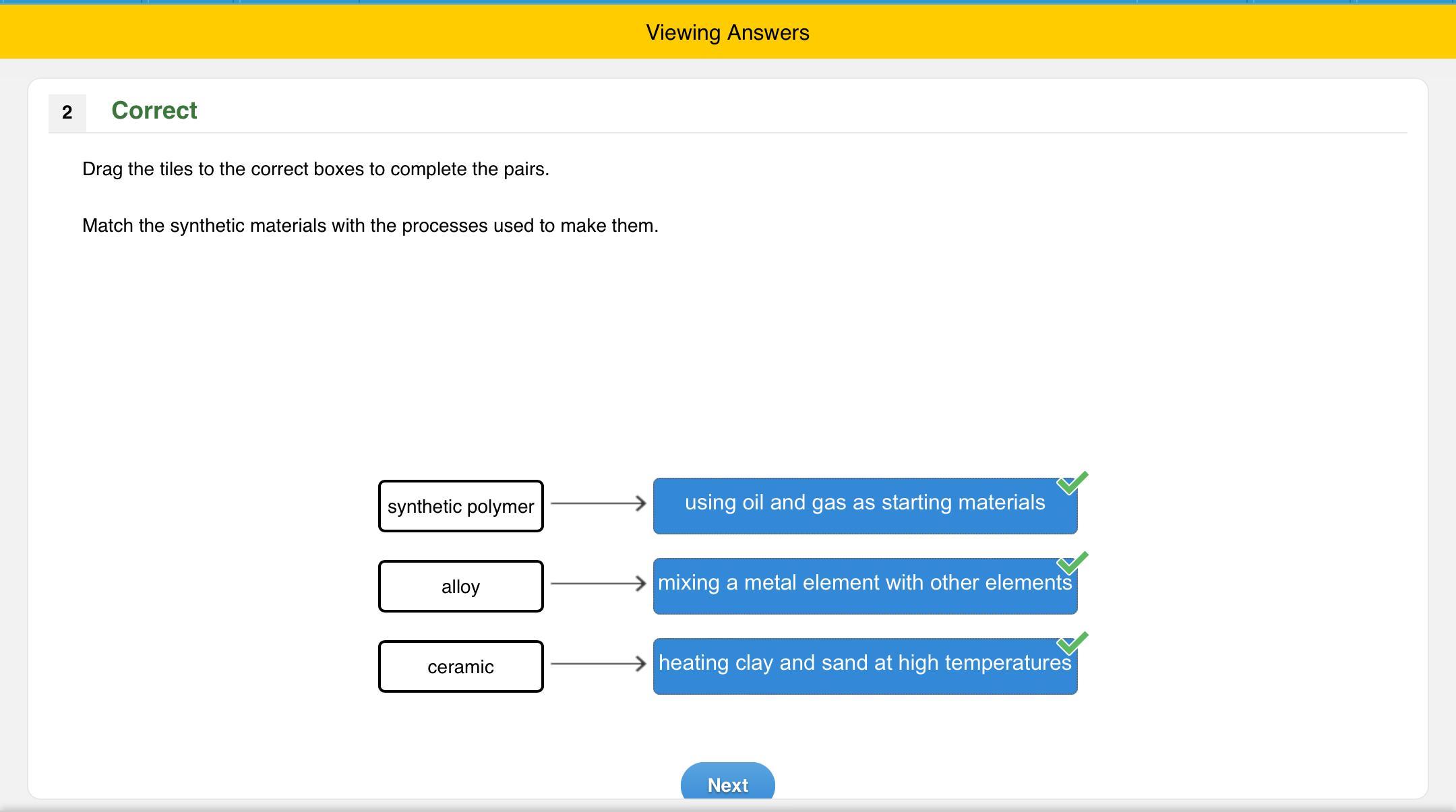
Drag the tiles to the correct boxes to complete the pairs match the synthetic materials with the processes used to make them.

Answers
Ceramic: A ceramic is any of the various hard, brittle, heat-resistant and corrosion-resistant materials made by shaping and then firing an inorganic, nonmetallic material, such as clay, at a high temperature. Common examples are earthenware, porcelain, and brick.
Synthetic polymer: Synthetic polymers are derived from petroleum oil, and made by scientists and engineers. Examples of synthetic polymers include nylon, polyethylene, polyester, Teflon, and epoxy. ... Vulcanized rubber is a synthetic (man-made) polymer, while pectin is an example of a natural polymer
That’s a *hint*
Answer:
I hope this helps
Explanation:
Related Questions
how many millimeters of a 0.266 M LiNO3 solution are required to make 150 mL of 0.075 M LiNO3 solution
Answers
Total, 42.37 millimeters of the 0.266 M Lithium nitrate solution are required to make 150 mL of the 0.075 M LiNO₃ solution.
To determine the volume of a 0.266 M LiNO₃ solution required to make 150 mL of a 0.075 M LiNO₃ solution, we can use the equation:
C₁V₁ = C₂V₂
Where;
C₁ = initial concentration of the LiNO₃ solution (0.266 M)
V₁ = volume of the LiNO₃ solution to be measured in millimeters (mm)
C₂ = final concentration of the LiNO₃ solution (0.075 M)
V₂ = final volume of the LiNO₃ solution (150 mL)
Rearranging the equation, we have;
V₁ = (C₂V₂) / C₁
Substituting the given values;
V₁ = (0.075 M) × (150 mL) / (0.266 M)
Converting mL to mm (1 mL = 1 mm), we get:
V₁ = (0.075 M) × (150 mm) / (0.266 M)
Calculating the result;
V₁ ≈ 42.37 mm
Therefore, approximately 42.37 millimeters of the 0.266 M LiNO₃ solution are required to make 150 mL of the 0.075 M LiNO₃ solution.
To know more about Lithium nitrate here
https://brainly.com/question/26319767
#SPJ4
How long will hydrogen peroxide make a dog throw up?.
Answers
Question 14 of 34
for a reaction, ah = 2 kj. for which value of tas is the reaction
spontaneous?
oa. -2 kj
b. 3 kj
oc. 2 kj
od. -3 kj
Answers
Option B is correct answer, when TΔS is 3 KJ the reaction is spontaneous reaction.
What is spontaneous reaction?
A spontaneous process in thermodynamics is one that happens without the system receiving any outside input. The time-evolution of a system in which it releases free energy and transitions to a lower, more thermodynamically stable energy state is a more technical definition (closer to thermodynamic equilibrium). Following the general convention for thermodynamic measurements, the sign convention for changes in free energy is as follows: a release of free energy from the system corresponds to a negative change in the system's free energy and a positive change in the surrounding free energy.
The reaction's spontaneity is determined by the Gibbs Free Energy (ΔG) value.
This value is calculated using the following equation, which depicts the link between temperature, entropy (ΔS), enthalpy (ΔH), and the Gibbs free energy (ΔG):
ΔG = ΔH - T(ΔS)
A positive ΔG value represents a non-spontaneous reaction,
A negative ΔG value represents a spontaneous reaction.
Given ΔH = 2 KJ,
We get ΔG negative only when TΔS is 3 KJ.
To know more about spontaneous reaction, go to link
https://brainly.com/question/24376583
#SPJ4
Even before modern observations provided evidence supporting the theory of plate tectonics, people developed theories that the continents were once joined together. Using only maps, what did they observe?
Answers
Answer:
people or scientist tend to observe the nature of the continents that described by jigsaw fit,geological similarity among the continents
Three solutions have pH values of 6, 7, and 11. How much more acidic is the pH 6 solution than the pH 7 solution if
acidity is defined as [H3O+]? How much more basic is the pH 11 solution
than the pH 7 solution if basicity is defined as [OH-]?
Answers
The solution at pH 6 is 10 times more acidic than the solution at pH 7.
The solution at pH 11 is 40 times more basic than the solution at pH 7.
What is the pH of the solution?Hydronium ion concentration at pH 8 = \(1 * 10^-6\)M
Hydronium ion concentration of pH 7 = \(1 * 10^-7\) M
Thus we have;\(1 * 10^-6 M/1 * 10^-7 M\) = 10
At pH 7, pOH = 7 and hydroxide ion concentration = \(1 * 10^-7\) M
At pH 11, pOH = 3 and the hydroxide ion concentration = \(1 * 10^-3\) M
We then have that; \(1 * 10^-3 M/ 1 * 10^-7\) M = 40
Learn more about pH:https://brainly.com/question/15289741
#SPJ1
Classify the following substances as strong electrolytes, weak electrolytes, or nonelectrolytes:
(b) Potassium nitrate (KNO₃)
Answers
\(KNO3\) is a strong electrolyte.
What is electrolyte.Substance that conducts electric current as a result of a dissociation into positively and negatively charged particle called ions, which migrate toward and ordinarily are discharged at the negative and positive terminals (cathode and anode) of an electric circuit, respectively.
To know more about the electrolyte visit :
https://brainly.com/question/28305069
#SPJ4
What property of matter will keep your body in motion when the car comes to halt
Answers
Answer:
Inertia in our body tends to be in the situation it is. So, we a car comes to a halt, our body wants to keep moving i.e. in motion. hope that helps love!
Which equation is correct?
A. TC + or - WCA = TH.
B. TH + or - DEV = MH.
C. MH + or - VAR = CH.
Answers
The equation that is correct is TC + or - WCA = TH. The correct option is
A.
The given equations are related to the magnetic compass errors.
The full form for the above equation is as follows :
T is the concerning TRUE North it is use to plotter and the latitude
M is Regarding the MAGNETIC North that is the True North +/- Deviation.
C is to the COMPASS North that is the Magnetic North +/- Compass card deviation in the system
C is Plotted COURSE in the map that is no wind correction is applied.
The order is as :
TC = True course
VAR = Variation
MC = Magnetic course
WCA = Wind correction angle
MH = Magnetic heading
DEV = Deviation
CH = Compass heading
To learn more about magnetic compass here
https://brainly.com/question/29870686
#SPJ4
identify the first step in preparing a spectrophotometer for use.
Answers
We have that the first step is to put the Power source into an on state,Thereby powering the Light point and the Spectrophotometer.
From the question we are told
identify the first step in preparing a spectrophotometer for use.
Generally
A SpectrophotometerThis is a device is out there to help scientist i the mostly in the field of chemistry.
This device is used to Know or arcertain particle with light consuming properties.
The Spectrophotometer is Mostly found in laboratories.
And usually in the use of a Spectrophotometer the first step is to put the Power source into an on state.Thereby powering the Light point and the Spectrophotometer
Therefore
The first step is to put the Power source into an on state,Thereby powering the Light point and the Spectrophotometer.
For more information on this visit
https://brainly.com/question/14379882
Enter a complete ionic equation to show the reaction of aqueous Hg2(NO3)2 with aqueous sodium chloride to form solid Hg2Cl2 and aqueous sodium nitrate. Express your answer as a complete ionic equation. Identify all of the phases in your answer.
Answers
Answer: \(Hg_2^{2+}(aq)+2NO_3^-(aq)+2Na^+(aq)+2Cl^-(aq)\rightarrow Hg_2Cl_2(s)+2Na^+(aq)+2NO_3^-(aq)\)
Explanation:
A double displacement reaction is one in which exchange of ions take place. The salts which are soluble in water are designated by symbol (aq) and those which are insoluble in water and remain in solid form are represented by (s) after their chemical formulas.
A double displacement reaction in which one of the product is formed as a solid is called as precipitation reaction.
The balanced chemical equation is:
\(Hg_2(NO_3)_2(aq)+2NaCl(aq)\rightarrow Hg_2Cl_2(s)+2NaNO_3(aq)\)
The complete ionic equation will be :
\(Hg_2^{2+}(aq)+2NO_3^-(aq)+2Na^+(aq)+2Cl^-(aq)\rightarrow Hg_2Cl_2(s)+2Na^+(aq)+2NO_3^-(aq)\)
3. Nicotine, the addictive drug in cigarettes, contains 74.0% carbon, 8.6% hydrogen,
and 17.3% nitrogen. What mass of each element can be recovered from a 55.0-
gram sample of nicotine?
Answers
Answer:
the mass of each element can be recovered from is 162.23 grams
Explanation:
the mass really depends on the volume. If there is 74.0% carbon, 8.6% hydrogen and 17.3% nitrogen then you first cross out the 0 and 3 which leaves you with 17% and 74%. Then all you have to do is multiply 17% and 74% and then divided it by 8.6%.
The various drugs such as the nicotine as additive drug is a pack of cigarette consist of about 75 of the carbon, 8.6% of hydrogen and about 17.3% of mitogen.
The M of each elements that can recovered which is 162.23 gM really depends on the volume, then you 1st cross out the 0 and 3 which leaves you with 17% and 74%. Then we have to do is multiply 17% and the 74% and divided it by the 8.6%.Learn more about the addictive drug.
brainly.com/question/2210950.
which one is a Metalloid (semimetal):
germanium
zinc
chlorine
Answers
Answer:
it is germanium
Explanation:Germanium is a chemical element with the symbol Ge and atomic number 32. It is a lustrous, hard-brittle, grayish-white metalloid in the carbon group, chemically similar to its group neighbours silicon and tin. Pure germanium is a semiconductor with an appearance similar to elemental silicon
The one that is a metalloid is germanium. The correct option is A.
What is a metalloid?A metalloid is a type of chemical element that has properties that are intermediate between those of metals and nonmetals.
There is no universal definition of a metalloid, and there is no universal agreement on which elements are metalloids.
Metalloid, an imprecise term used in chemistry to describe a chemical element that forms a simple substance with properties intermediate between those of a typical metal and those of a typical nonmetal.
Boron (86), silicon (95), germanium (96), arsenic (100), selenium (23), antimony (88), and astatine are the elements with the highest percentage appearance frequencies as metalloids (40).
Thus, the correct option is A.
For more details regarding metalloids, visit:
https://brainly.com/question/2548493
#SPJ6
If a star is 300 light years away from Earth, how long does it take light from that star to reach Earth?
A. 300 days
B. 300 hours
C. 300 years
D. 300 light years
Answers
Answer: It would take 300 light years to reach Earth.
Explanation:
PLS FAST WILL GIVE BRAINLIEST!!! Rocky road ice cream is an example of a _____________________ mixture.
Answers
Answer:
Rocky road ice cream is an example of a __________Homogeneous___________ mixture.
Atomic #
Mass #
#p
#e
#n
charge
Symbol
1) 17
19
0
2)
180
71
109
3)
40
38
46
4) 92
238
86
206
5)
Pb4
82
6)
34
45
-2
7)
113
48
49
8) 21
42
0
31
9)
p.
15
10)
83
80
126

Answers
In the modern periodic table, the elements are listed in order of increasing atomic number. The atomic number is the number of protons in the nucleus of an atom. The number of protons define the identity of an element (i.e., an element with 6 protons is a carbon atom, no matter how many neutrons may be present).
which are true about lubricants for cutting? they are composed almost entirely of hydrocarbons with some additives. they should be used at lower cutting speeds than other types of cutting fluids. they make cutting surfaces smoother. they dissolve easily in water they make it easier for cutting chips to not to stick to the cutting tool. they reduce the temperature of the cutting tool, but not as much as other cutting fluids.
Answers
A lubricant must keep its properties in the presence of additives and other items. Lubricating oil colours are more of a test for a grade or brand of oil's consistency than they are for its attributes. Thus, option B is correct.
What are the characteristics of lubricants?There are four different kinds of liquid lubricants. Mineral oil a. It is a material made from petroleum. These oils are stable at high temperatures because they have a high paraffine and naphthanic content.
Examples include steam cylinder oil, wire rope oil, refrigeration grade oil, gear oil, machine or engine oil, and circulating oil.
Therefore, they should be used at lower cutting speeds than other types of cutting fluids. They make cutting surfaces smoother.
Learn more about lubricants here:
https://brainly.com/question/30082267
#SPJ4
In what areas of the country should this material be marked?
Answers
Answer:
Explanation: The goal of every country should be to produce more materials and goods
What is the molecular formula of a compound with an empirical formula CHOCl and a molecular weight of 129 g
Answers
The molecular formula of the compound is C2H2O2Cl2.
To determine the molecular formula of a compound with the empirical formula CHOCl and a molecular weight of 129 g, we need to find the actual number of atoms of each element in the compound.The empirical formula CHOCl suggests that the compound contains one carbon (C), one hydrogen (H), one oxygen (O), and one chlorine (Cl) atom.To calculate the molecular formula, we need to compare the empirical formula's empirical mass to the compound's actual molecular weight. The empirical mass of CHOCl can be calculated by adding the atomic masses of the constituent elements: C (12.01 g/mol) + H (1.01 g/mol) + O (16.00 g/mol) + Cl (35.45 g/mol) = 64.47 g/mol.By dividing the molecular weight of 129 g by the empirical mass of 64.47 g/mol, we find that the compound's molecular formula is approximately C2H2O2Cl2.The molecular formula C2H2O2Cl2 indicates that the compound contains two carbon atoms, two hydrogen atoms, two oxygen atoms, and two chlorine atoms. This formula has a molecular weight of approximately 129 g, which matches the given molecular weight.
for such more questions on molecular
https://brainly.com/question/24191825
#SPJ8
How does changing the electronegativity of the atoms affect the bond polarity?.
Answers
The polarity of a bond can be defined as the degree to which the electrons in a chemical bond are distributed between the two atoms of the bond. Polarity arises from differences in electronegativity, which is a measure of the ability of an atom to attract electrons.
Therefore, changes in electronegativity have an impact on bond polarity. When two atoms with different electronegativities bond, the electrons are attracted to the more electronegative atom. This results in the more electronegative atom having a partial negative charge, and the less electronegative atom having a partial positive charge.
This is known as a polar covalent bond. The greater the difference in electronegativity between the two atoms, the more polar the bond. If the difference in electronegativity between the two atoms is zero, the bond is considered nonpolar. Therefore, as the electronegativity of the atoms in a bond increases, the bond polarity increases.
To know more about bond visit:
https://brainly.com/question/31994049
#SPJ11
Please help me answer this it’s due today. I will give brainliest

Answers
Answer: X should represent H, hydrogen.
Explanation:
The H is the only one that hasnt been stated in the left side of the formula. H has three atoms as well.
In the electroplating of nickel, 0.200 faraday of electrical charge is passed through a solution of NiSO4. What mass of nickel is deposited?
a. 2.94
b. 5.87
c. 11.7
d. 58.7
Answers
When a solution of NiSO4 is exposed to a 0.2 faraday electrical charge, b. 5.87 g of nickel, Ni is deposited.
The Balanced equation is :
Ni²⁺ + 2e —> Ni
To Determine the mass of the Ni when there is 0.2 farad.
According to the balanced equation,
The molar mass of nickel is 1× 59 \(gmol^{-1}\)
The number of farad =2
(Since, In the balanced equation nickel losses 2 electron. )
2 farad = 59 \(gmol^{-1}\) of nickel
0.2 farad = ?
⇒\(\frac{(59)(0.2)}{2}\)
⇒5.87 \(gmol^{-1}\) ≈ 5.9 \(gmol^{-1}\)
What is Nickel Electroplating ?Nickel is deposited onto a metal component by the technique of nickel electroplating. Before plating can start, parts must be clean and free of debris, corrosion, and flaws. A mixture of heat treating, cleaning, masking, pickling, and etching may be used to prepare and protect the part before plating.
To know more about Nickel Electroplating refer here
https://brainly.com/question/23612460#
#SPJ4
If an object has a mass of 30 g and a volume of 2 cm ^ 3 , what would its density ?
Answers
Answer:
The answer is
15.0 g/cm³Explanation:
The density of a substance can be found by using the formula
\(density = \frac{mass}{volume} \\ \)
From the question
mass of object = 30 g
volume = 2 cm³
The density of the object is
\(density = \frac{30}{2} \\ \)
We have the final answer as
15.0 g/cm³Hope this helps you
Which things below would help make an ecosystem more biodiverse? O Having a very large population of a species O Having a large community of organisms O Lots of different traits in a species Not having seasons
Answers
• An ecosystem refers to multiple species
• A large population of one species is a decent answer but not the best because more species would have more diverse genetics
• Lots of traits in one species is basically the same as the above bullet point
• Not having seasons could help but not necessarily
• So our best answer is a large community of organisms
•
help me balence: ___ H2 + ___ O2----> _____ H20
Answers
Explanation:
2H2+O2------->2H2O
its yr balanced equation.
hope it helps
stay safe healthy and happy....in a 78.0-g 78.0 -g aqueous solution of methanol, ch4o, ch 4 o , the mole fraction of methanol is 0.100. 0.100. what is the mass of each component?
Answers
The mole fraction of methanol is 0.100 in a 78.0-g 78.0-g aqueous solution of methanol, ch4o, ch 4 o. Is the mass of methanol in the solution is 7.809 g and the mass of water is 39.425 g.
The mole fraction of methanol (CH4O) in the solution is 0.100, which means that 10% of the moles of the solution are methanol. We can use this information, along with the total mass of the solution, to determine the mass of each component.
The total moles of the solution can be calculated by dividing the total mass of the solution by its molar mass. Methanol has a molar mass of 32.04 g/mol, so we have:
total moles of solution = 78.0 g / 32.04 g/mol = 2.434 mol
Now we can calculate the moles of methanol:
moles of methanol = 0.100 x 2.434 mol = 0.2434 mol
The remaining moles are water:
moles of water = 2.434 mol - 0.2434 mol = 2.1906 mol
To calculate the mass of each component, we can use their respective molar masses:
mass of methanol = 0.2434 mol x 32.04 g/mol = 7.809 g
mass of water = 2.1906 mol x 18.02 g/mol = 39.425 g
Therefore, the mass of methanol in the solution is 7.809 g and the mass of water is 39.425 g.
Learn more about molar mass here:
https://brainly.com/question/12127540
#SPJ4
6. Look carefully at the following diagram. It does not obey the law of conservation of matter. Explain why it does not follow the law of conservation of matter using evidence
from the diagram.

Answers
Answer:
Sort of answer, but it'll lead you in the right direction I hope.
Explanation:
I mean, ofc, the molecules or atoms or whatever the circles are can't be put together that way bc matter says so. It looks like the other diagram is showing water or gas, and if it were turning into the one on the right, then it would usually either settle into water if it was gas or become a single solid object like water. If this is gas, then it doesn't make sense, because gas particles are always separate from each other.
Nuclear symbol notation for hydrogen-1
Answers
Answer:
1H, 2H, and 3H.
Explanation:
secondary alcohol (X) reacts with PCL5 to give alkyl halide (Y) which on dehydrohalogenation yield an alkene(Z).The alkene upon ozonolysis gives the mixture of ethanal and methanal.Identify X ,Y ,Z
Answers
Answer:
X = 2-propanol
Y = 2-chloropropane
Z = propene
Explanation:
Secondary alcohol is an alcohol in which the carbon that the functional group (OH group) is attached to is also attached to two alkyl group. Examples are 2-propanol and 2-butanol.
The secondary alcohol referred to in the question here is 2-propanol and it reacts with PCl₅ as seen below
H₃C-CH-CH₃ + PCl₅ ⇒ H₃C-CH-CH₃ + HCl + POCl₃
| |
OH Cl
The alkylhalide produced there is 2-chloropropane. Thus, we can see that X is 2-propanol and Y is 2-chloropropane.
When this 2-chloropropane undergoes dehydrohalogenation (removal of hydrogen and halogen) an alkene is formed. The reaction is seen below
H₃C-CH-CH₃ (-HCl) ⇒ H₃C-CH=CH₂
|
Cl
The alkene produced (as seen above) is propene.
Confirmation: When propene undergoes ozonolysis, the compounds produced are ethanal and methanal as seen in the equation below
O
/ \
H₃C-CH=CH₂ + O₃ ⇒ H₃C - CH CH₂ [-O] ⇒ CH + CH₂
|| || || ||
O O O O
The final products as seen above are methanal and ethanal
using the standard enthalpies of formation, what is the standard enthalpy of reaction?
Answers
Answer:
This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. and the standard enthalpy of formation values: ΔH fo[A] = 433 KJ/mol.
Explanation:
Hope this helps.
Explain the reason why you tested electrical conductivity with a solution containing both methanol and water.
Answers
In comparison to non-polar hydrocarbon dielectrics like gasoline and diesel fuel, methanol is a polar substance like water and has a relatively high conductivity (conductivities less than 1 x 10⁴ pS/m).
Does methanol have a higher conductivity than water?Therefore, the self dissociation into ions for methanol and ethanol will be less strong than in water. But even in pure ethanol and methanol solutions, these ions will still exist and conduct electricity, albeit at very low conductivities.
Water's electrical conductivity is a measure of how well it can carry a current of electricity. The amount of electrical current that can be conducted in water increases with the concentration of dissolved charged chemicals, also referred to as salts.
To know more about electrical conductivity visit:
https://brainly.com/question/29765686
#SPJ4